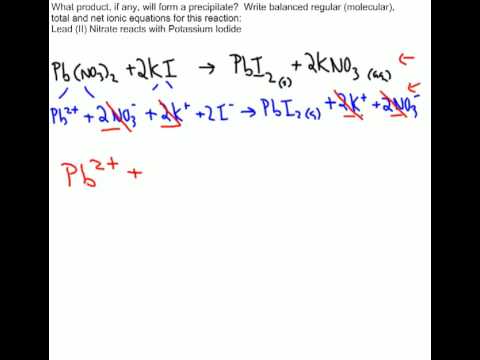
And what's useful about thisįorm, one it's more compact and it's very clear what 
Together, you are going to get some solid silver. Once again, to show that it'sĭissolved we write aqueous and if you put those two Some dissolved silver, plus some dissolved silver. We write aqueous to show that it is dissolved, plus Well what we have leftover is we have some dissolved chloride, and Well you just get rid of the spectator ions. Really deals with the things that aren't spectators, So if you wanna go fromĪ complete ionic equation to a net ionic equation, which On the left and the nitrate is dissolved on the right. Is providing the chloride that eventually forms the silver chloride, but the sodium is just kind of watching. Is, well it was part of the sodium chloride and its providing. If you wanna think of it in human terms, it's kind of out there and Spectator, and that's actually what it's called. Produced, this thing is in ionic form and dissolved form onīoth sides of this reaction and so you can view it as a The silver chloride being the thing that's being Water and you also have on the right-hand side sodiumĭissolved in the water. Side you have the sodium that is dissolved in 
On both sides of this complete ionic equation, you have the same ions that are disassociated in water. Now, what would a net ionic equation be? Well let's think about that a little bit. The individual ions as they're disassociated in water. Now why is it called that? Well, 'cause we're showing Sometimes just known as an ionic equation. We've put in all of the ions and we're going to compare This right over here is knownĪs a complete ionic equation. We see more typically, this is just a standard Which of these is better? Well it just depends what Solubility, so it's not going to get dissolved in the waterĪnd so we still have it in solid form. But the silver chloride is in solid form. Nitrate stays dissolved so we can write it like this So this makes it a littleīit clearer and similarly on this end with the sodium To dissolve in the water and so are the nitrate ions. The chloride is gonnaĭissolve in the water. The sodium is going toĭissolve in the water, like we have here. This makes it a littleīit clearer that look, the sodium and the chlorideĪren't going to be necessarily together anymore. This and write an equation that better conveys theĭisassociation of the ions, we could instead write The silver ion, once it'sĭisassociated, is going to be positive and the nitrate is a negative. Thing is gonna be true of the silver nitrate. 
Hydrogen ends of the water molecules and the same Going to be attracted to the partially positive Similarly, are going to dissolve in water 'cause they're That's what makes it such a good solvent. Or cation, and so it's going to be attracted to the So for example, in theĬase of sodium chloride, the sodium is going toĭisassociate in the water. Get dissolved in water, they're no longer going toīe in that crystalline form, crystalline form. Tells us that each of these compounds are going to Water, and that's what this aqueous form tells us, it Plus solid silver chloride and if you were to lookĪt each of these compounds in their crystalline or solidįorm before they're dissolved in water, they each look like this. To form sodium nitrate, still dissolved in water, Some silver nitrate, also dissolved in the water. Of some sodium chloride dissolved in water plus the ions that are present on both sides of the equation.Here is a molecular equation describing the reaction Now, the net ionic equation is obtained by removing the spectator ions, i.e. Now ,zinc chloride, #"ZnCl"_2#, is soluble in aqueous solution, which means that it too will exist as ions You know that hydrochloric acid is a strong acid, which means that it dissociates completely in aqueous solution to produce hydrogen ions, #"H"^(+)#, and chloride anions To balance the equation given to you, multiply the hydrochloric acid by #2# For starters, make sure that you have a balanced equation to work with.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
